There are many substances in the Universe. Wood, metal, glass, dirt, flesh, and so forth. All of which are made of atoms. These substances often change forms through various processes known as physical and chemical changes.
Physical changes are changes that happen to a substance without changing its chemical composition. For example, if you melt an ice cube, it becomes water. The ice cube is still made of water molecules, just in a different form. Other examples of physical changes include cutting, grinding, and boiling.
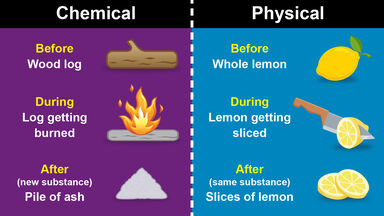
Chemical changes are changes that actually change the chemical composition of a substance. For example, when you burn wood, the molecules in the wood change into other molecules like carbon dioxide and water vapor. This is a chemical change because the wood is now made of different molecules than it was before. Other examples of chemical changes include rusting and fermentation.
Physical and chemical changes are alike in that they both change the form or composition of a substance. However, physical changes do not change the chemical composition of a substance, while chemical changes do.
Examples of physical changes include :
– Melting
– Freezing
– Cutting
– Grinding
– Boiling
– Evaporation
– Condensation
– Sublimation
Examples of chemical changes include:
– Burning
– Rusting
– Fermentation
– Combustion
– Decomposition